Effect of vitamin D on endothelial and ventricular function in chronic heart failure patients
- Jan 5, 2023
- 1 min read
Medicine (Baltimore) 2022 Jul 22;101(29):e29623.
Jong Shin Woo, Yeongmin Woo, Jeong Yoon Jang, Sang Jin Ha
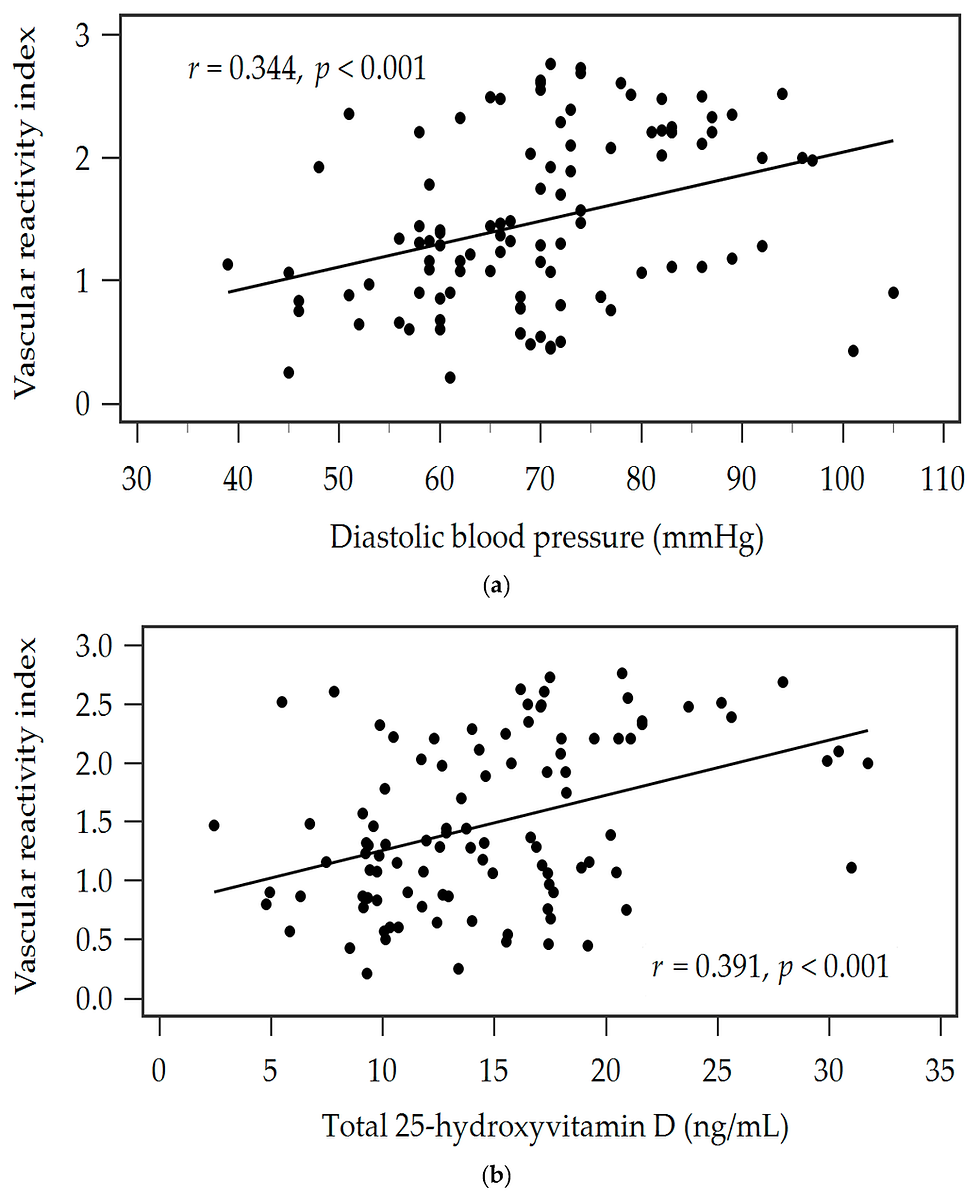
Abstract Background: Low 25-hydroxyvitamin D (25OHD) levels are common in patients with chronic heart failure (HF) and are associated with increased mortality risk. This study aimed to establish the safety and efficacy of oral vitamin D3 (cholecalciferol) supplementation and its effect on endothelial and ventricular function in patients with stable HF. Methods: This study was an investigator-initiated, multicenter, prospective, randomized, placebo-controlled trial. Seventy-three HF patients with 25OHD levels < 75 nmol/L (30 ng/mL) were randomized to receive 4000 IU vitamin D daily or a placebo for 6 months. The primary endpoint was a change in endothelial function between the baseline and after 6 months as assessed using EndoPAT. Secondary endpoints included changes in echocardiographic parameters and differences in quality of life (6-min walking test and New York Heart Association functional status) at 6 months.
Results: There were no adverse events in either group during the study period. Vitamin D supplementation did not improve endothelial dysfunction (EndoPAT: baseline, 1.19 ± 0.4 vs 6 months later, 1.22 ± 0.3, P = .65). However, patients' blood pressure, 6-min walking distance, and EQ-5D questionnaire scores improved after vitamin D treatment. In addition, a significant reduction in the left atrial diameter was observed.
Conclusion: A daily vitamin D dose of 4000 IU for chronic HF appears to be safe. This dosage did not improve endothelial function but did improve the 6-min walk distance, symptoms, and left atrial diameter at 6 months.
View Full Text Here: https://journals.lww.com/md-journal/Fulltext/2022/07220/Effect_of_vitamin_D_on_endothelial_and_ventricular.45.aspx
Copyright © 2022 the Author(s). Published by Wolters Kluwer Health, Inc.



Comments